We improve
quality of life
by creating new
medicines
News
10 years of the NTI NeuroNet Recreation Center: from hypotheses to a sustainable industry and technological independence in Russia

Chronic viral hepatitis C: innovations in therapy

How to get a world-class high-tech product for the pharmaceutical industry discussed at the XII Congress «Innovative Practice: Science plus Business»

The work of the NTI TDK expert group was highly appreciated by the ASI leadership and recognized with “People Change the Country” award

Domestic pharmaceutical industry “get stuck” on generics

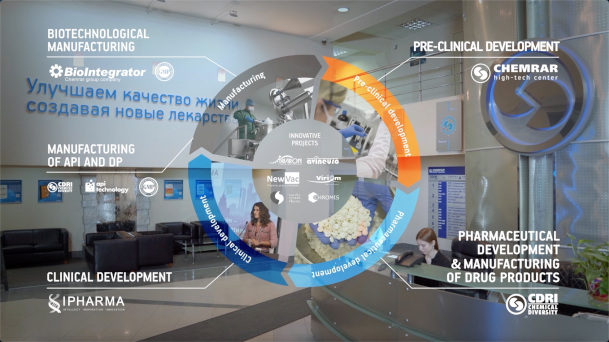
Infrastructure & Facilities
ChemRar Research and Development Institute
Vivarium and biochemical laboratories (GLP)
Chemical Diversity Research Institute
Production of innovative drugs and generics (GMP)
BioIntegrator
Production of biotechnological medicines and vaccines
API-Technology
Production of pharmaceuticals ingredients (GMP)
ChemRar vision & mission
We develop and manufacture effective, safe and affordable medicines for the treatment of socially significant diseases and improvement of the quality of life:
- Using experience and global standards at all stages of creation of a novel medicine;
- Contributing to the development of the scientific and technological potential of the pharmaceutical industry in Russia;
- Applying our knowledge to search for optimal solutions for our partners.
We develop and manufacture effective, safe and affordable medicines for the treatment of socially significant diseases and improvement of the quality of life:
- Using experience and global standards at all stages of creation of a novel medicine;
- Contributing to the development of the scientific and technological potential of the pharmaceutical industry in Russia;
- Applying our knowledge to search for optimal solutions for our partners.
Product pipeline
Avineuro

Avineuro is engaged in research and development of original drugs for the treatment of CNS disorders
Viriom

Viriom company at ChemRar High-Tech Center develops innovative antiviral drugs and combinations, methods of treatment and diagnosis
CardioSystem
Pharma
Pharma

CardioSystemPharma has developed and is launching on the Russian market a new cardioplegic drug, Normacor, for open-heart surgery at a normal body temperature
Aviron

The Aviron project of ChemRar Group implements a program for the development of innovative antiviral drugs with different complementary mechanisms of action for the treatment of chronic hepatitis C, which is included in the list of socially significant diseases and known as “the silent killer”.
NewVac

NewVac is engaged in the research and development of personalized and combination approaches to cancer therapy.
Chromis

The Russian Direct Investment Fund (RDIF), Russia’s sovereign wealth fund, and the ChemRar Group launched JV Chromis for production of antiviral drug Avifavir based on Favipiravir.